Subscribe to RSS

DOI: 10.1055/s-0039-3402490
Chordoid Meningioma: Literature Review
Meningioma cordoide: Revisão de literaturaAuthors
Address for correspondence
Publication History
02 April 2019
22 October 2019
Publication Date:
11 March 2020 (online)
Abstract
Chordoid meningiomas (CMs) are a rare subgroup of tumors, accounting for ∼ 0.5% of all meningiomas. Chordoid meningioma tumors correspond to World Health Organization (WHO) Grade II lesions and behave aggressively, with an increased likelihood of recurrence. There are few genetic studies about CMs, but we understand that there is deletion at many chromosomal loci. Histologically, CMs are characterized by strands and cords of meningothelial cells arranged in a mucinous stroma. Morphologically, it can mimic other chondroid and myxoid tumors within the brain and its vicinity, thus posing a diagnostic challenge. Chordoid meningiomas have an aggressive clinical course and a propensity to recur compared with classical meningiomas. The goal of the treatment is surgery, with total resection of the tumor; however, due to its high degree of recurrence, radiotherapy is often necessary as an adjuvant treatment.
Resumo
Meningiomas cordoides (MCs) são um tipo raro de subgrupo de tumores, representando ∼ 0,5% de todos os meningiomas. Os MCs correspondem ao grau II, pela classificação da Organização Mundial da Saúde (OMS), possuindo agressividade e alto grau de recorrência. Existem poucos estudos publicados sobre a genética desses tumores; porém, entendemos que há diversas deleções em alguns lócus cromossômicos. Histologicamente, os MCs são caracterizados por fios e cordões de células meningoteliais dispostos em um estroma mucinoso. Morfologicamente, podem mimetizar outros tumores condroides e mixoides, o que representa um desafio diagnóstico. Os MCs têm um curso mais agressivo que o meningioma clássico. O objetivo do tratamento é a cirurgia, com ressecção total do tumor, porém, devido à sua alta recidiva, se faz necessário o tratamento adjuvante com radioterapia.
Introduction
Chordoid meningioma (CM) is a rare variant of meningioma that bears a striking histological resemblance to chordoma and has a greater likelihood of recurrence. The differential diagnosis includes chordomas, myxoid chondrosarcoma and chordoid glioma.[1] In the present study, we report a case of a female patient with a diagnosis of a recent onset of headache and visual cloudiness. Meningiomas represent almost 30% of primary intracranial neoplasms.
Chordoid meningioma is a rare subtype of meningioma, accounting to 0.5% of all meningiomas and associated with different behavior.[2]
Chordoid meningiomas are classified in Grade II by the World Health Organization (WHO).[3] Chordoid meningioma has a similar distribution between men and women. Chordoid meningioma can have a large distribution in the age range, and may range from 4 to 77 years old.[4] A little more than 100 cases of chordoid meningioma have been described in the English language literature, the majority of which are in the pathology and neurosurgery literature.
Material and Methods
Case Report
Female, 42-year-old, with complaints of sudden onset of headache and visual impairment. Upon neurological examination, the patient presented paresis of the left medial rectus muscle. The neurosurgery team requested a magnetic resonance imaging (MRI) exam, which showed an expansive lesion in the left frontotemporal topography. The lesion was causing an important compression of the medial rectus muscle ([Fig. 1]). We opted for a left frontotemporal craniotomy, since it was possible to resect the lesion. The patient progressed well during surgery and, after the postoperative period, was discharged from the hospital feeling well and with no neurological deficits. ([Fig. 2])


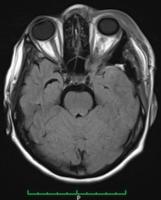

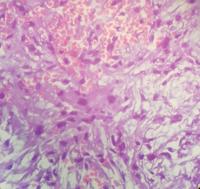
We sent the material for pathological analysis, which revealed mesenchymal neoplasia with chordoid pattern in the left temporal frontographic topography ([Figs. 1] and [2]). Therefore, the diagnosis was of chordoid meningioma. The immunohistochemistry analysis was positive for epithelial membrane antigen, therefore being typical of the chordoid meningioma ([Figs. 3] and [4]). The literature review involved clinical case reports, randomized controlled trials and a series of cases describing clinical and epidemiological treatments for chordoid meningioma. The search was performed using the PubMed database and targeting all English language publications available involving chordoid meningioma. This review was organized applying the MeSH terms chordoid meningioma, meningioma and chordoid. The search about chordoid meningioma at PubMed/Medline retrieved 83 articles. For all these articles, we applied filters such as species (humans) and age (19 to 65 years old). Then, we retrieved 59 papers. Analyzing the titles of the articles, 59 were selected for summary review, considering relevance and relation to the present paper. Among those 59, 31 were selected and examined.



Results
Meningiomas have preponderance for convexities. For instance, 17 to 25% occur in a frontobasal location. Within the frontobasal region, the olfactory grooves, the tuberculum sellae, the parasellar region and the petrous bone are preferred sites.[5] On the other hand, 5% occur along the cerebellar convexity, 2 to 4% at the tentorium cerebelli, and 2 to 4% within the cerebellopontine angle (CPA).[6]
Chordoid meningioma is an uncommon histopathological variant of meningioma frequently mistaken as chordoma, due to its histological similarity. Couce et al related 42 clinicopathologic studies about CM during the period of 1975 to 1997, and this study concluded that it was more common in the supratentorial region and in adults.[6] [7]
There are only two studies about genetic alterations in CMs. The majority of meningiomas is known to have deletion at many chromosomal loci not yet studied in CMs, such as 22q, 18p, 14q and 1p. All of the literature cases showed to have either complete or partial deletion at 22q, 14q and 1p loci.[8] [9] Additionally, all cases were variably positive for epithelial membrane antigen (EMA) and showed strong positive staining for vimentin, but were completely negative for glial fibrillary acidic protein (GFAP), in support of the diagnosis.[9] Because of the similarity between CMs and other lesions, especially tumors, careful assessment of each case is required. The main diagnoses are chondroid choroid, chordoid glioma, paraganglioma, metastatic mucinous carcinoma or metastatic renal cell carcinoma, among others.[10]
The goal of the therapy in CM is relieving the compression of critical anatomic structures as well as preventing tumor spread and recurrence.[11] The therapeutic preference for aggressive meningiomas are surgical resection, radiotherapy (RT) and chemotherapy, in cases of anaplastic meningioma.[10] [11] Neurosurgery is the treatment of choice for meningiomas and, preferentially, with total resection.[10]
Discussion
Meningiomas are the second most common central nervous system neoplasm in adults and account for 15 to 20% of all primary brain tumors. Meningiomas are benign tumors; however 10% of the cases demonstrate a more aggressive clinical behavior, such as chordoid meningioma, which is a type of non-benign meningioma.[12] First described by Kepes in 1988, chordoid meningiomas are characterized by cords of eosinophilic, epithelial or spindle-shaped cells within a myxoid stroma, resembling chordoma. Variable numbers of lymphocytes and plasma cells characterize these tumors.
Chordoid meningiomas occur very rarely, corresponding to up to 1% of surgically removed meningiomas, even in large cohorts. Chordoid meningiomas are located supratentorially in 81.5% of the cases.[13] Unusual sites, such as the ventricular system, the foramen jugulare and the orbital area have been described in several case reports.[14] [15] Histologically, CM consists of cords or trabeculae of eosinophilic and vacuolated cells embedded in an abundant mucoid matrix.[16]
However, sometimes the histological diagnosis is difficult because it is comparable morphology in other intracranial tumors, such as chordoid glioma, chordoma, extraskeletal chondrosarcoma, myxopapillary ependymoma and metastatic tumors.[8]
Immunohistopathological studies have shown that this tumor shows positivity for vimentin, EMA and D2–40, and occasional positivity for S-100 protein, on the other hand demonstrating negativity for GFAP and cytokeratin.[9] [10] [11] [12] [13] [14] [15] [16] [17] [18] [19]
The first choice of treatment for CM is surgical excision. The goal of surgery is a complete resection. The complete resection of a tumor is sometimes difficult because of its location.[8] [9] [10] [11] The surgical strategy is to perform total resection of the tumor, dura and bone, while protecting the surrounding structures. In contrast, when an incomplete removal is realized, due to the risk of injury to the surrounding vital neurovascular structures, postoperative adjuvant RT is an acceptable option.[16] [19] [20] Despite this recommendation, there is controversy surrounding the value of postoperative RT in atypical meningiomas. This treatment is commonly recommended for patients with atypical meningiomas following subtotal removal, but some investigators, acknowledging the greater recurrence risk of atypical meningiomas, recommend early irradiation, irrespective of the resection extent, to optimize local control and improve survival.[5] Once recurrent, grade II meningiomas carry a mortality risk and postoperative RT, after total removal, may prolong or prevent recurrence.[9] Perhaps, more comprehensive series are required to evaluate the role of brain invasion and postoperative RT in the prognosis of CM.[1] [2] [3] [4] [5] [6] [7] [8] [9] [10] [11] [12] [13] [14] [15] [16] [17] [18] [19] [20] [21] In our case, we reconvened the patient to continue the treatment and improve the prognosis.
Conclusion
Chordoid meningiomas are rare and different tumors of the meningiomas that present a high degree of recurrence, even when the satisfactory resection of the lesion is achieved. Recurrence is particularly worrisome after a subtotal surgical resection. Their histological similarity to chordoid neoplasms, especially chordoma, can make the diagnosis challenging. These two neoplasms have different biological behaviors, so it is important to be able to distinguish them. The goal of treatment is surgery, with total resection of the tumor. In cases of relapse or in cases of residual tumor, it is necessary to perform RT.
Conflict of Interests
The authors have no conflict of interests to declare.
Acknowledgments
The authors would like to thank the patient for her cooperation and also the staff of the hospital.
-
Reference
- 1 Jee TK, Jo KI, Seol HJ, Kong DS, Lee JI, Shin HJ. Clinical features and treatment outcome of chordoid meningiomas in a single institute. J Korean Neurosurg Soc 2014; 56 (03) 194-199
- 2 Sriram PR. Chordoid meningioma, part of a multiple intracranial meningioma: a case report & review. Malays J Med Sci 2013; 20 (04) 91-94
- 3 Presta I, Guadagno E, Di Vito A. , et al. Innate immunity may play a role in growth and relapse of chordoid meningioma. Int J Immunopathol Pharmacol 2017; 30 (04) 429-433
- 4 Di Ieva A, Laiq S, Nejad R. , et al. Chordoid meningiomas: incidence and clinicopathological features of a case series over 18 years. Neuropathology 2015; 35 (02) 137-147
- 5 Civit T, Baylac F, Taillandier L, Auque J, Hepner H. [Chordoid meningiomas. Clinical, neuroradiological and anatomopathological aspects. Apropos of a new case and review of the literature]. Neurochirurgie 1997; 43 (05) 308-313
- 6 Zhang GJ, Zhang YS, Zhang GB. , et al. Prognostic Factors, Survival, and Treatment for Intracranial World Health Organization Grade II Chordoid Meningiomas and Clear-Cell Meningiomas. World Neurosurg 2018; 117: e57-e66
- 7 Couce ME, Aker FV, Scheithauer BW. Chordoid meningioma: a clinicopathologic study of 42 cases. Am J Surg Pathol 2000; 24 (07) 899-905
- 8 Siraj F, Ansari MK, Sharma KC, Singh A. Chordoid meningioma: A diagnostic dilemma. J Cancer Res Ther 2015; 11 (03) 663
- 9 Sugur H, Shastry AH, Sadashiva N, Srinivas D, Santosh V, Somanna S. Chromosomal aberrations in chordoid meningioma - An analysis. Neurol India 2018; 66 (01) 156-160
- 10 Malloy KA, Chigbu DI. Anterior temporal chordoid meningioma causing compressive optic neuropathy. Optom Vis Sci 2011; 88 (05) 645-651
- 11 Choy W, Ampie L, Lamano JB. , et al. Predictors of recurrence in the management of chordoid meningioma. J Neurooncol 2016; 126 (01) 107-116
- 12 Sadashiva N, Poyuran R, Mahadevan A, Bhat DI, Somanna S, Devi BI. Chordoid meningioma: a clinico-pathological study of an uncommon variant of meningioma. J Neurooncol 2018; 137 (03) 575-582
- 13 Passacantilli E, Lapadula G, Caporlingua F. , et al. Chordoid meningioma: a retrospective series of seven consecutive cases. Neurol Sci 2013; 34 (11) 1985-1989
- 14 Kozler P, Benes V, Netuka D, Kramár F, Hrabal P, Charvát F. Chordoid meningioma: presentation of two case reports, review of the literature, and plea for data standardisation. J Neurooncol 2008; 88 (01) 115-120
- 15 Epari S, Sharma MC, Sarkar C, Garg A, Gupta A, Mehta VS. Chordoid meningioma, an uncommon variant of meningioma: a clinicopathologic study of 12 cases. J Neurooncol 2006; 78 (03) 263-269
- 16 Jiménez-Heffernan JA, Urquía-Renke A, Bárcena C, Fraga J. Utility of diff-quik stained smears in the cytologic diagnosis of chordoid meningioma. Diagn Cytopathol 2016; 44 (10) 811-813
- 17 McIver JI, Scheithauer BW, Atkinson JL. Deep Sylvian fissure chordoid meningioma: case report. Neurosurgery 2005; 57 (05) E1064 , discussion E1064
- 18 Lin JW, Lu CH, Lin WC. , et al. A clinicopathological study of the significance of the proportion of choroid morphology in chordoid meningioma. J Clin Neurosci 2012; 19 (06) 836-843
- 19 Pond JB, Morgan TG, Hatanpaa KJ, Yetkin ZF, Mickey BE, Mendelsohn DB. Chordoid Meningioma: Differentiating a Rare World Health Organization Grade II Tumor from Other Meningioma Histologic Subtypes Using MRI. AJNR Am J Neuroradiol 2015; 36 (07) 1253-1258
- 20 Yang Y, Li D, Cao XY. , et al. Clinical Features, Treatment, and Prognostic Factors of Chordoid Meningioma: Radiological and Pathological Features in 60 Cases of Chordoid Meningioma. World Neurosurg 2016; 93: 198-207
- 21 Wang XQ, Mei GH, Zhao L. , et al. Clinical features and treatment of intracranial chordoid meningioma: a report of 30 cases. Histopathology 2013; 62 (07) 1002-1017
Address for correspondence
-
Reference
- 1 Jee TK, Jo KI, Seol HJ, Kong DS, Lee JI, Shin HJ. Clinical features and treatment outcome of chordoid meningiomas in a single institute. J Korean Neurosurg Soc 2014; 56 (03) 194-199
- 2 Sriram PR. Chordoid meningioma, part of a multiple intracranial meningioma: a case report & review. Malays J Med Sci 2013; 20 (04) 91-94
- 3 Presta I, Guadagno E, Di Vito A. , et al. Innate immunity may play a role in growth and relapse of chordoid meningioma. Int J Immunopathol Pharmacol 2017; 30 (04) 429-433
- 4 Di Ieva A, Laiq S, Nejad R. , et al. Chordoid meningiomas: incidence and clinicopathological features of a case series over 18 years. Neuropathology 2015; 35 (02) 137-147
- 5 Civit T, Baylac F, Taillandier L, Auque J, Hepner H. [Chordoid meningiomas. Clinical, neuroradiological and anatomopathological aspects. Apropos of a new case and review of the literature]. Neurochirurgie 1997; 43 (05) 308-313
- 6 Zhang GJ, Zhang YS, Zhang GB. , et al. Prognostic Factors, Survival, and Treatment for Intracranial World Health Organization Grade II Chordoid Meningiomas and Clear-Cell Meningiomas. World Neurosurg 2018; 117: e57-e66
- 7 Couce ME, Aker FV, Scheithauer BW. Chordoid meningioma: a clinicopathologic study of 42 cases. Am J Surg Pathol 2000; 24 (07) 899-905
- 8 Siraj F, Ansari MK, Sharma KC, Singh A. Chordoid meningioma: A diagnostic dilemma. J Cancer Res Ther 2015; 11 (03) 663
- 9 Sugur H, Shastry AH, Sadashiva N, Srinivas D, Santosh V, Somanna S. Chromosomal aberrations in chordoid meningioma - An analysis. Neurol India 2018; 66 (01) 156-160
- 10 Malloy KA, Chigbu DI. Anterior temporal chordoid meningioma causing compressive optic neuropathy. Optom Vis Sci 2011; 88 (05) 645-651
- 11 Choy W, Ampie L, Lamano JB. , et al. Predictors of recurrence in the management of chordoid meningioma. J Neurooncol 2016; 126 (01) 107-116
- 12 Sadashiva N, Poyuran R, Mahadevan A, Bhat DI, Somanna S, Devi BI. Chordoid meningioma: a clinico-pathological study of an uncommon variant of meningioma. J Neurooncol 2018; 137 (03) 575-582
- 13 Passacantilli E, Lapadula G, Caporlingua F. , et al. Chordoid meningioma: a retrospective series of seven consecutive cases. Neurol Sci 2013; 34 (11) 1985-1989
- 14 Kozler P, Benes V, Netuka D, Kramár F, Hrabal P, Charvát F. Chordoid meningioma: presentation of two case reports, review of the literature, and plea for data standardisation. J Neurooncol 2008; 88 (01) 115-120
- 15 Epari S, Sharma MC, Sarkar C, Garg A, Gupta A, Mehta VS. Chordoid meningioma, an uncommon variant of meningioma: a clinicopathologic study of 12 cases. J Neurooncol 2006; 78 (03) 263-269
- 16 Jiménez-Heffernan JA, Urquía-Renke A, Bárcena C, Fraga J. Utility of diff-quik stained smears in the cytologic diagnosis of chordoid meningioma. Diagn Cytopathol 2016; 44 (10) 811-813
- 17 McIver JI, Scheithauer BW, Atkinson JL. Deep Sylvian fissure chordoid meningioma: case report. Neurosurgery 2005; 57 (05) E1064 , discussion E1064
- 18 Lin JW, Lu CH, Lin WC. , et al. A clinicopathological study of the significance of the proportion of choroid morphology in chordoid meningioma. J Clin Neurosci 2012; 19 (06) 836-843
- 19 Pond JB, Morgan TG, Hatanpaa KJ, Yetkin ZF, Mickey BE, Mendelsohn DB. Chordoid Meningioma: Differentiating a Rare World Health Organization Grade II Tumor from Other Meningioma Histologic Subtypes Using MRI. AJNR Am J Neuroradiol 2015; 36 (07) 1253-1258
- 20 Yang Y, Li D, Cao XY. , et al. Clinical Features, Treatment, and Prognostic Factors of Chordoid Meningioma: Radiological and Pathological Features in 60 Cases of Chordoid Meningioma. World Neurosurg 2016; 93: 198-207
- 21 Wang XQ, Mei GH, Zhao L. , et al. Clinical features and treatment of intracranial chordoid meningioma: a report of 30 cases. Histopathology 2013; 62 (07) 1002-1017












