Subscribe to RSS
Please copy the URL and add it into your RSS Feed Reader.
https://www.thieme-connect.de/rss/thieme/en/10.1055-s-00000083.xml
Synlett 2016; 27(14): 2128-2132
DOI: 10.1055/s-0035-1561473
DOI: 10.1055/s-0035-1561473
cluster
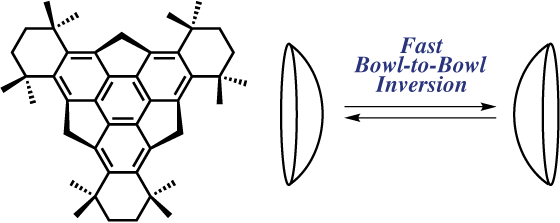
Bowl-to-Bowl Inversion Accelerated by the Introduction of Bulky 1,1,4,4-Tetramethylbutane-1,4-diyl Groups on Sumanene
Further Information
Publication History
Received: 08 April 2016
Accepted after revision: 15 May 2016
Publication Date:
22 June 2016 (online)

Abstract
A variable-temperature 1H NMR study revealed that the bowl-to-bowl inversion of sumanene is accelerated by the introduction of bulky 1,1,4,4-tetramethylbutane-1,4-diyl groups on the peripheral aromatic ring. This is likely to be induced by a bowl flattening, as shown in the DFT-optimized structures.
Key words
bowl-to-bowl inversion - sumanene - bowl-shaped polyaromatic molecule - Friedel–Crafts alkylation - variable-temperature 1H NMRSupporting Information
- Supporting information for this article is available online at http://dx.doi.org/10.1055/s-0035-1561473.
- Supporting Information
Primary Data
- Primary data for this article are available online at http://www.thieme-connect.com/products/ejournals/journal/10.1055/s-00000083 and can be cited using the following DOI: 10.4125/pd0079th.
- Primary Data
-
References and Notes
- 1 Scott LT, Hashemi MM, Bratcher MS. J. Am. Chem. Soc. 1992; 114: 1920
- 2 Sygula A, Abdourazak AH, Rabideau PW. J. Am. Chem. Soc. 1996; 118: 339
- 3 Seiders TJ, Baldridge KK, Grube GH, Siegel JS. J. Am. Chem. Soc. 2001; 123: 517
- 4a Sastry GN, Jemmis ED, Mehta G, Shah SR. J. Chem. Soc., Perkin Trans. 2 1993; 1867
- 4b Priyakumar UD, Sastry GN. J. Phys. Chem. A 2001; 105: 4488
- 5 Sakurai H, Daiko T, Hirao T. Science 2003; 301: 1878
- 6 Amaya T, Sakane H, Muneishi T, Hirao T. Chem. Commun. 2008; 765
- 7 Higashibayashi S, Sakurai H. J. Am. Chem. Soc. 2008; 130: 8592
- 8 Amaya T, Nakata T, Hirao T. J. Am. Chem. Soc. 2009; 131: 10810
- 9 Amaya T, Wang W.-Z, Sakane H, Moriuchi T, Hirao T. Angew. Chem. Int. Ed. 2010; 49: 403
- 10 Tsuruoka R, Higashibayashi S, Ishikawa T, Toyota S, Sakurai H. Chem. Lett. 2010; 39: 646
- 11 Amaya T, Nakata T, Sakane H, Hirao T. Pure Appl. Chem. 2010; 82: 965
- 12 Higashibayashi S, Tsuruoka R, Soujanya Y, Purushotham U, Sastry GN, Seki S, Ishikawa T, Toyota S, Sakurai H. Bull. Chem. Soc. Jpn. 2012; 85: 450
- 13 Amaya T, Hirao T. Pure Appl. Chem. 2012; 84: 1089
- 14 Tan Q, Higashibayashi S, Karanjit S, Sakurai H. Nat. Commun. 2012; 3: 891
- 15 Shrestha BB, Karanjit S, Higashibayashi S, Sakurai H. Pure Appl. Chem. 2014; 86: 747
- 16 Amaya T, Ito T, Hirao T. Eur. J. Org. Chem. 2014; 3531
- 17 Shrestha BB, Karanjit S, Higashibayashi S, Amaya T, Hirao T, Sakurai H. Asian J. Org. Chem. 2015; 4: 62
- 18 Amaya T, Ito T, Katoh S, Hirao T. Tetrahedron 2015; 71: 5906
- 19 Preparation Procedure for 4 and 5 To a mixture of sumanene (1, 10 mg, 0.038 mmol) and AlCl3 (45.3 mg, 0.34 mmol) in dry CH2Cl2 (1 mL) was added 2,5-dichloro-2,5-dimethylhexane (31.7 mg, 0.17 mmol) under a nitrogen atmosphere at room temperature. The reaction mixture was stirred for 1 h. The reaction mixture was filtered through silica gel (eluent: CH2Cl2), and the solvent was evaporated in vacuo to give the crude product (17 mg) including 3, 4, and 5. They were separated by recycling preparative HPLC with the GPC columns (eluent: CHCl3, column: tandemly arranged JAIGEL-1H and JAIGEL-2H, Japan Analytical Industry Co., Ltd.). Compound 4: FT-IR (KBr): ν = 2925, 2867, 2956, 1457, 1361 cm–1. 1H NMR (400 MHz, toluene-d 8, –40 °C, ref.: δ = 2.08 ppm): δ = 7.10 (s, 2 H), 4.85 (d, J= 19.0 Hz, 1 H), 4.50 (d, J= 19.2 Hz, 2 H), 4.14 (d, J= 19.2 Hz, 1 H), 3.57 (d, J= 19.2 Hz, 2 H), 1.80–1.60 (m, 8 H), 1.55 (s, 6 H), 1.50 (s, 6 H), 1.29 (s, 6 H), 1.18 (s, 6 H) ppm; 13C NMR (100 MHz, CDCl3, room temperature, ref.: δ = 77.0 ppm): δ = 147.4, 146.8, 144.7, 143.9, 143.65, 143.56, 142.3, 142.1, 122.8, 47.4, 44.7, 2 × 36.7, 36.1, 36.0, 32.9, 32.8, 2 × 29.7 ppm. HRMS (MALDI TOF): m/z [M+] calcd for C37H40: 484.3125; found: 484.3106. Compound 5: FT-IR (KBr): ν = 2959, 2925, 2864, 1457, 1361 cm–1. 1H NMR (400 MHz, toluene-d 8, –60 °C, ref.: δ = 2.08 ppm): δ = 4.90 (d, J= 19.0 Hz, 3 H), 4.21 (d, J= 19.2 Hz, 3 H), 1.82–1.65 (m, 12 H), 1.60 (s, 9 H), 1.34 (s, 9 H) ppm. 13C NMR (100 MHz, toluene-d 8, –40 °C, ref.: δ = 20.4 ppm): δ = 143.9, 143.7, 141.6, 47.7, 36.8, 36.1, 32.9, 29.8 ppm. HRMS (MALDI TOF): m/z [M+] calcd for C45H54: 594.4220; found: 594.4237.
- 20 All calculations were conducted by using the Gaussian 09, Revision D.01 program suite: Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA. Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ. Gaussian 09, Revision D.01 Program Suite. Gaussian Inc; Wallingford, CT: 2013
- 21 Sakurai H, Daiko T, Sakane H, Amaya T, Hirao T. J. Am. Chem. Soc. 2005; 127: 11580
